Technical Specifications:
- Details
-
1.
Main technical and operational characteristics:
1.1.
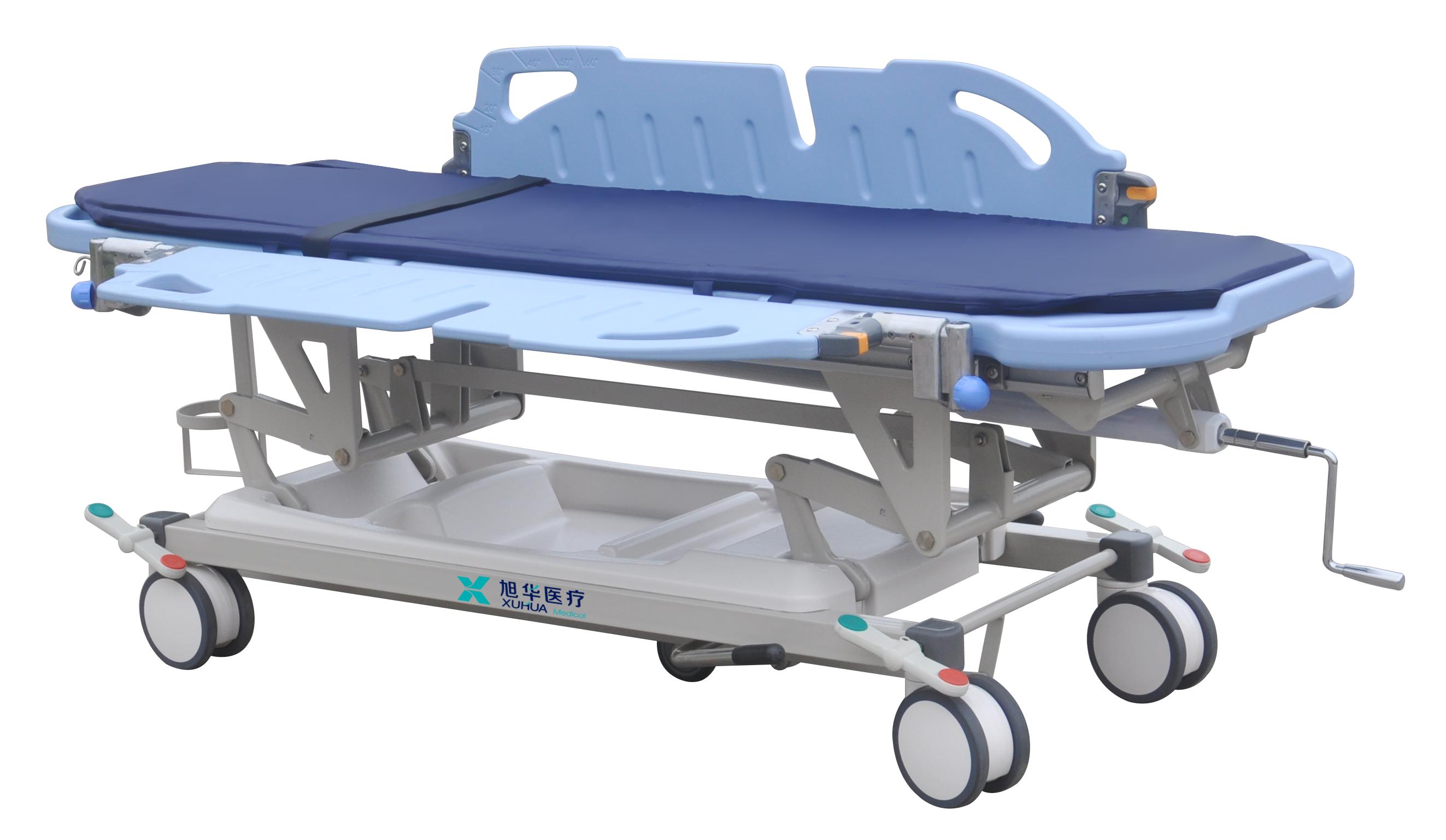
The bed is designed for use in maternity hospitals and perinatal centers to ensure optimal ergonomics and comfort for both the patient and medical staff throughout all stages of labor (LDR - Labor, Delivery, Recovery), as well as for various diagnostic, obstetric, and treatment procedures.
1.2.
The bed shall be of a transforming type (to accommodate various birthing positions) featuring a robust construction, modern ergonomic design, and user-friendly functionality. The frame and joints must exhibit high-quality craftsmanship, with seamless welding and superior surface finishing of all junctions.
1.3.
The bed frame shall be constructed of high-quality steel with an eco-friendly polymer coating, resistant to intensive treatment with hospital-grade disinfectants
1.4.
Electrically actuated 3-section configuration mattress platform comprising backrest, seat (pelvic), and foot sections.
1.5.
External size (L x W) : 1950 х 930 mm±50mm
1.6.
Height adjustment range: 690 - 900 mm±50mm
1.6.1.
Adjustment method - electric (via hand controller):
1.7.
Backrest adjustment range: 0° - 70°±5°
1.7.1.
Adjustment method - electric (via hand controller):
1.8.
Trendelenburg/rev-Trendelenburg range: ± 10°±2°
1.8.1.
Adjustment method - electric (via hand controller):
1.9.
Seat section (pelvic section) with perineal cutout:
1.10.
Leg section single-piece or split; removable (retractable leg section option is permissible):
1.11.
Mattresses - removable, multi-layer (polyurethane foam and viscoelastic foam), with pressure redistribution properties; thickness 140 mm:
1.12.
Mattress cover - polyurethane (waterproof, vapor-permeable, resistant to medical disinfectants); with zip closure:
1.12.1.
Technical information for the proposed mattress and cover:
1.13.
Safe working load: 200kg
1.14.
Bed mobility provided by 4 twin swivel (360°) casters:
1.14.1.
Caster diameter: 125mm
1.14.2.
Central braking system (single pedal operation to lock/unlock all 4 casters):
1.14.3.
Caster material - polyurethane (casters must be non-marking):
1.14.4.
Caster design - enclosed with plastic lateral covers (shrouds):
1.15.
Headboard - ABS plastic construction, removable:
1.16.
Siderails - ABS plastic construction, with soft-drop lowering mechanism:
1.17.
4-corner wall protection bumpers:
1.18.
Manual CPR release lever (for rapid emergency backrest lowering):
1.19.
Side accessory rails on both sides of the seat section (pelvic section) for attachment of various medical accessories:
1.20.
IV pole mounting receptacle:
1.21.
Angle indicators for mattress platform and backrest inclination:
2.
Power supply:
2.1.
220 V ± 10 %, 50/60 Hz:
2.2.
Mains power cable; EU standard plug (Schuko type):
3.
Additional equipment, components, spare parts, consumables:
3.1.
Removable stainless steel fluid basin, volume 10 liters:
3.1.1.
Basin design: retractable via stainless steel sliding rails under the seat section.
3.2.
Goepel type leg rests, removable: 1 pair
3.2.1.
Material - polyurethane:
3.2.2.
Ball joint clamp (articulated joint) for multi-directional positioning:
3.3.
Foot supports, removable: 1 pair
3.3.1.
Material - polyurethane (or polyurethane coating):
3.4.
Polyurethane-coated hand grips (labor grips) for assistance during second stage of labor: 1 pair
3.5.
Polyurethane-coated birthing bar (squat bar): 1 pc.
3.6.
Telescopic IV pole (4 hooks): 1 pc.
3.7.
Drainage bag holder: 2 pcs.
3.8.
Hand controller holder integrated into the bed frame: 1 pc.
3.9.
Accessory mounting mechanisms: 1 set
4
Warranty: 2 years
Previous Page
Next page
Previous Page
Next page
Related products
Online Message